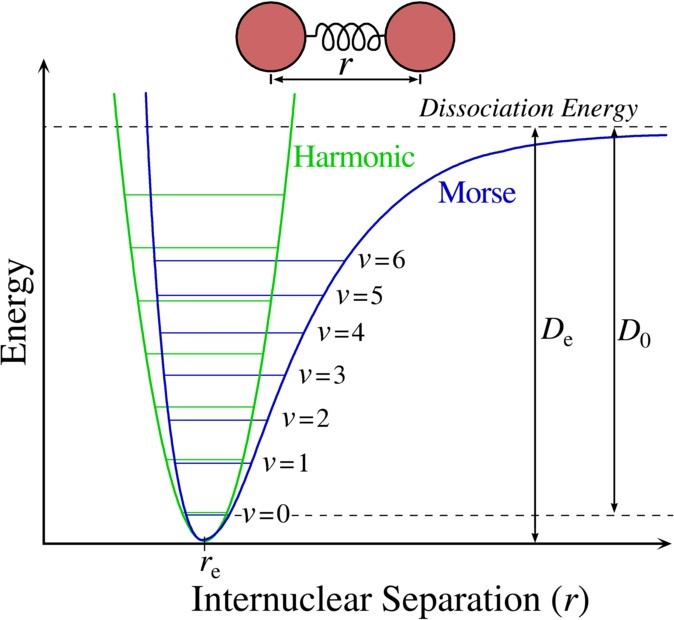
Communication: Determination of the bond dissociation energy (D0) of the water dimer, (H2O)2, by velocity map imaging: The Journal of Chemical Physics: Vol 134, No 21

Dissociating stable nitrogen molecules under mild conditions by cyclic strain engineering | Science Advances
Accurate Dissociation of Chemical Bonds Using DFT-in-DFT Embedding Theory with External Orbital Orthogonality

Density Functional Theory plus U Study of Methanol Adsorption and Decomposition on CuO Surfaces with Oxygen Vacancy - Aerosol and Air Quality Research

Exceptionally High O–H Bond Dissociation Free Energy of a Dicopper(II) μ-Hydroxo Complex and Insights into the Geometric and Electronic Structure Origins Thereof | Journal of the American Chemical Society

Benchmark DFT studies on C–CN homolytic cleavage and screening the substitution effect on bond dissociation energy | SpringerLink

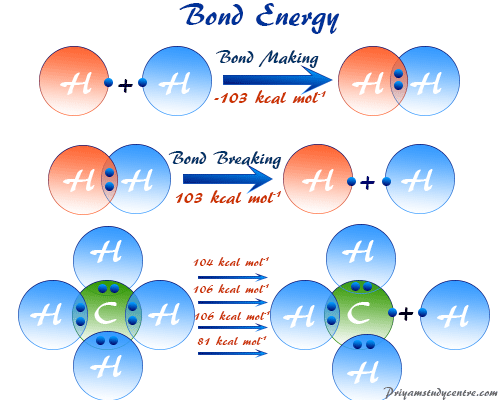
A level Bond Enthalpy (bond dissociation energy) calculations for Enthalpy of Reaction KS5 GCE chemistry revision notes
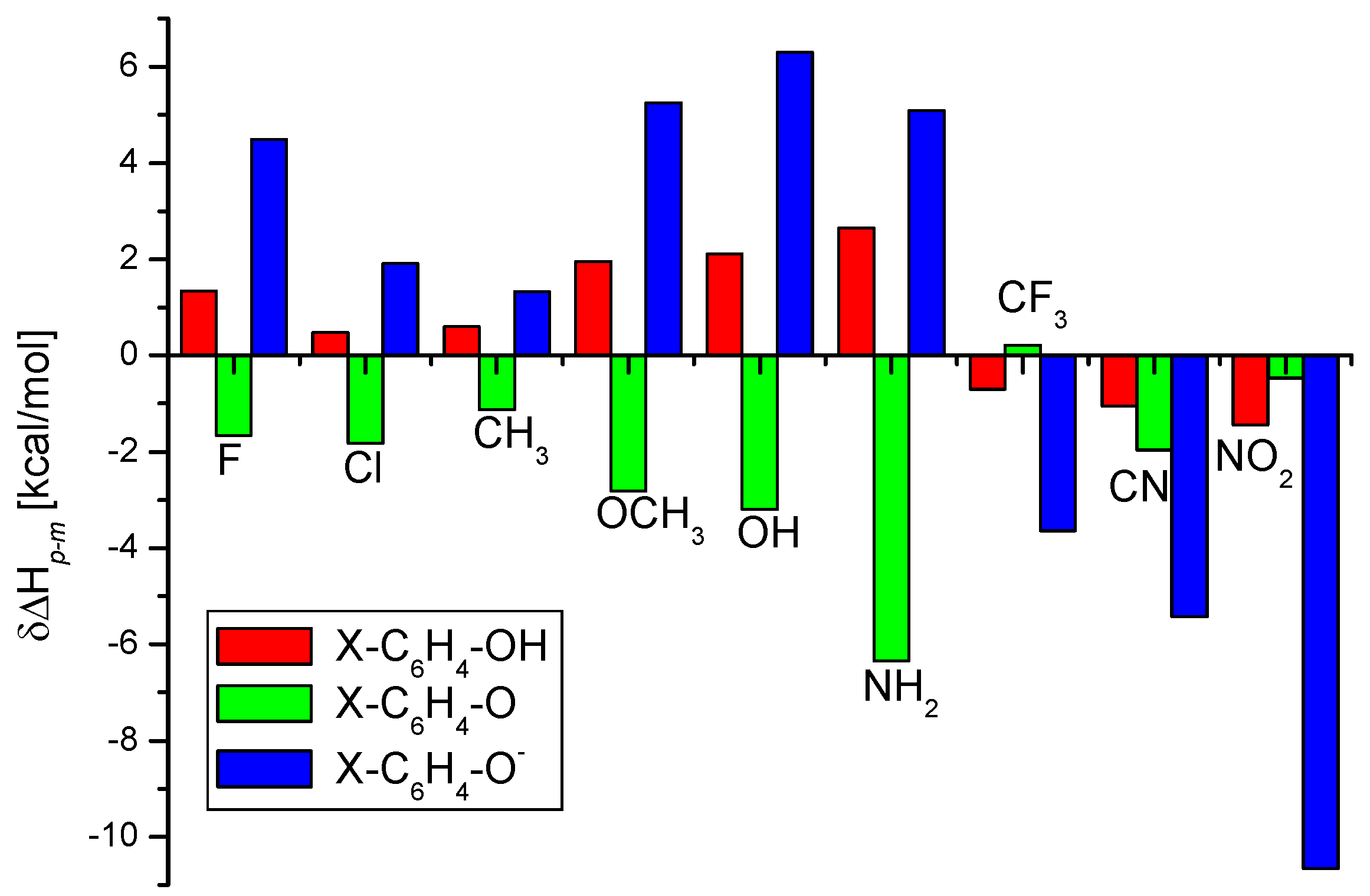
IJMS | Free Full-Text | The O-H Bond Dissociation Energies of Substituted Phenols and Proton Affinities of Substituted Phenoxide Ions: A DFT Study
Computation of the bond dissociation enthalpies and free energies of hydroxylic antioxidants using the ab initio Hartree–Fock

Benchmark study of bond dissociation energy of SiX (XF, Cl, Br, N, O, H and C) bond using density functional theory (DFT) - ScienceDirect
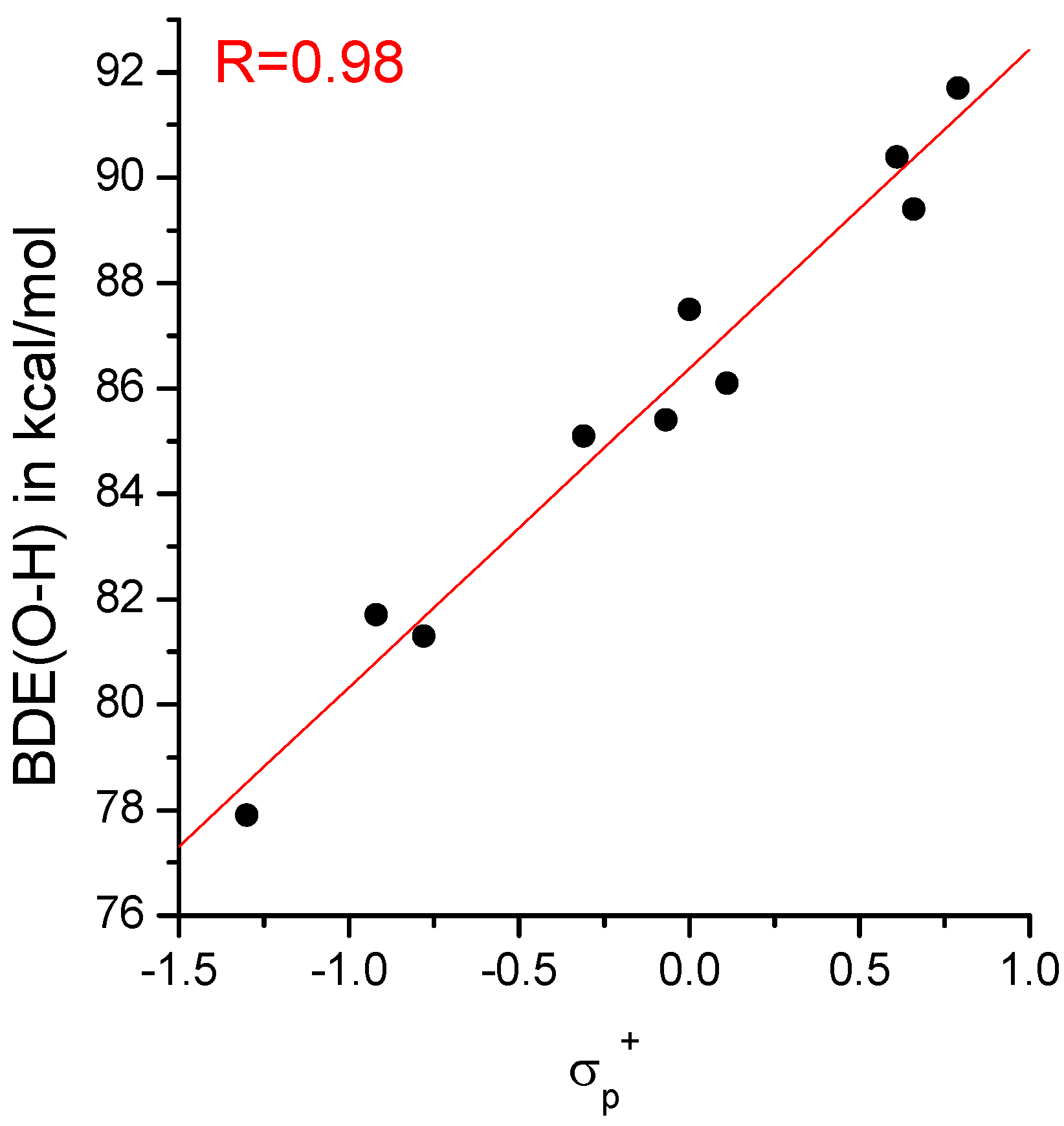
IJMS | Free Full-Text | The O-H Bond Dissociation Energies of Substituted Phenols and Proton Affinities of Substituted Phenoxide Ions: A DFT Study
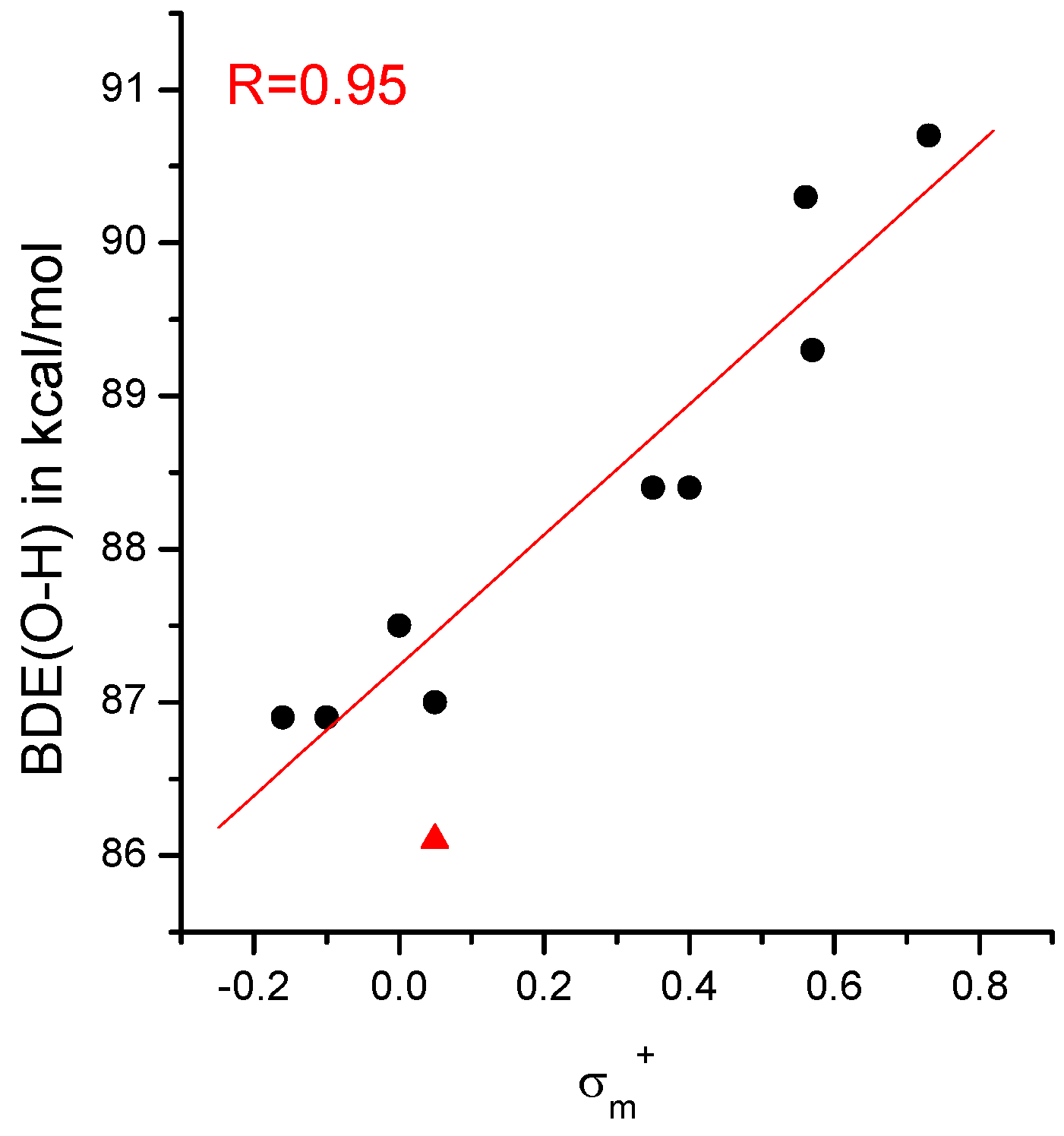
IJMS | Free Full-Text | The O-H Bond Dissociation Energies of Substituted Phenols and Proton Affinities of Substituted Phenoxide Ions: A DFT Study

Manipulating the Water Dissociation Electrocatalytic Sites of Bimetallic Nickel‐Based Alloys for Highly Efficient Alkaline Hydrogen Evolution - Wang - 2022 - Angewandte Chemie International Edition - Wiley Online Library

Water‐Assisted Chemical Route Towards the Oxygen Evolution Reaction at the Hydrated (110) Ruthenium Oxide Surface: Heterogeneous Catalysis via DFT‐MD and Metadynamics Simulations - Creazzo - 2021 - Chemistry – A European Journal -

Minimum energy difference calculated using DFT and ReaxFF of (a) bond... | Download Scientific Diagram

Density Functional Theory Study of Water Molecule Adsorption on the α-Quartz (001) Surface with and without the Presence of Na+, Mg2+, and Ca2+ | ACS Omega

High-throughput DFT calculations of formation energy, stability and oxygen vacancy formation energy of ABO3 perovskites | Scientific Data
Benchmark calculations for bond dissociation energies and enthalpy of formation of chlorinated and brominated polycyclic aromatic hydrocarbons - RSC Advances (RSC Publishing)

![PDF] Bond dissociation energies of organic molecules. | Semantic Scholar PDF] Bond dissociation energies of organic molecules. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3aaecb82c2ece7f77647ce525290bf3d9d4eb750/2-Table1-1.png)