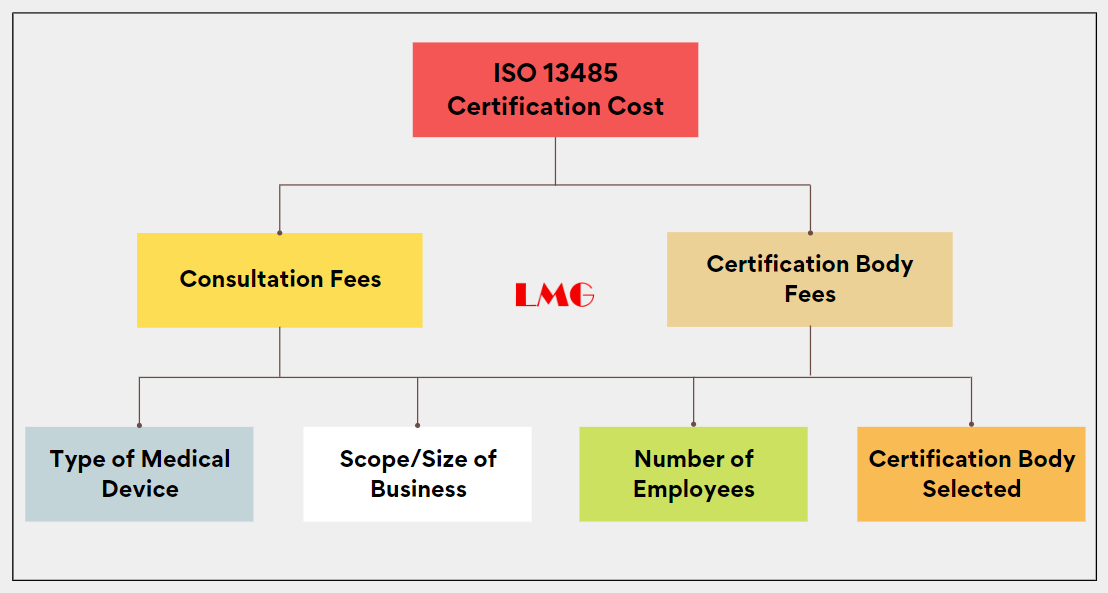
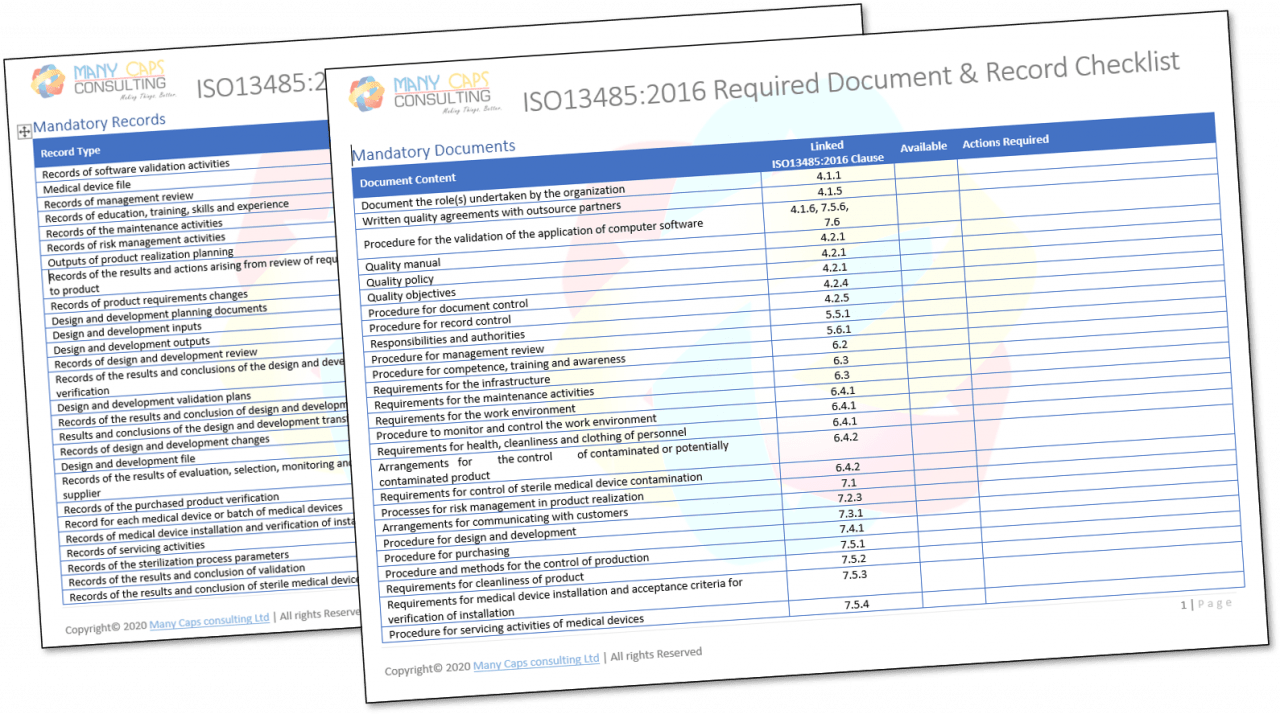
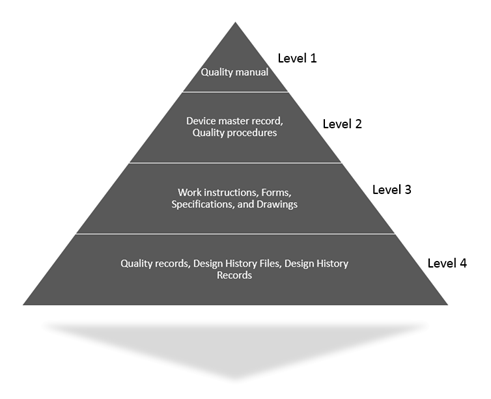
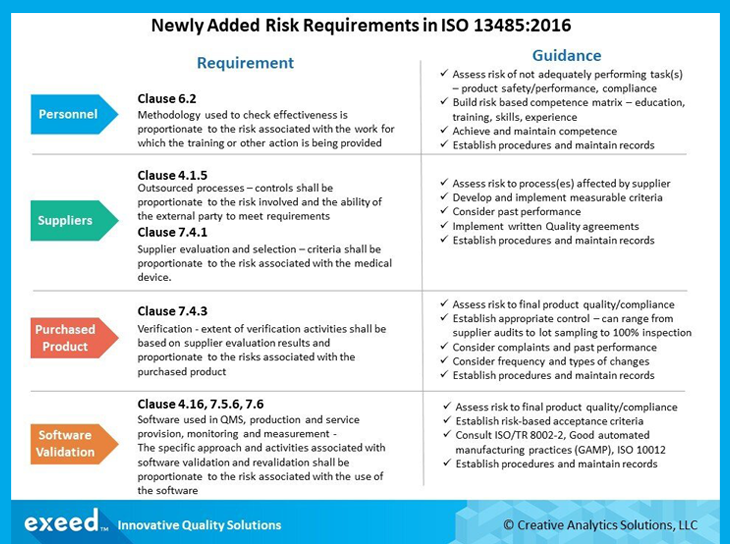
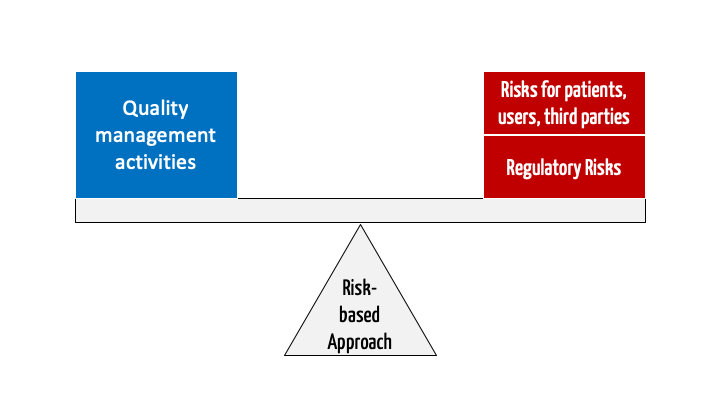
ISO 13485 – Medical Device Quality Management System Requirements – ISO Templates and Documents Download

Poor Quality of ISO Documents | Cost of Quality Calculation Example | ISO 9001 Quality System - YouTube
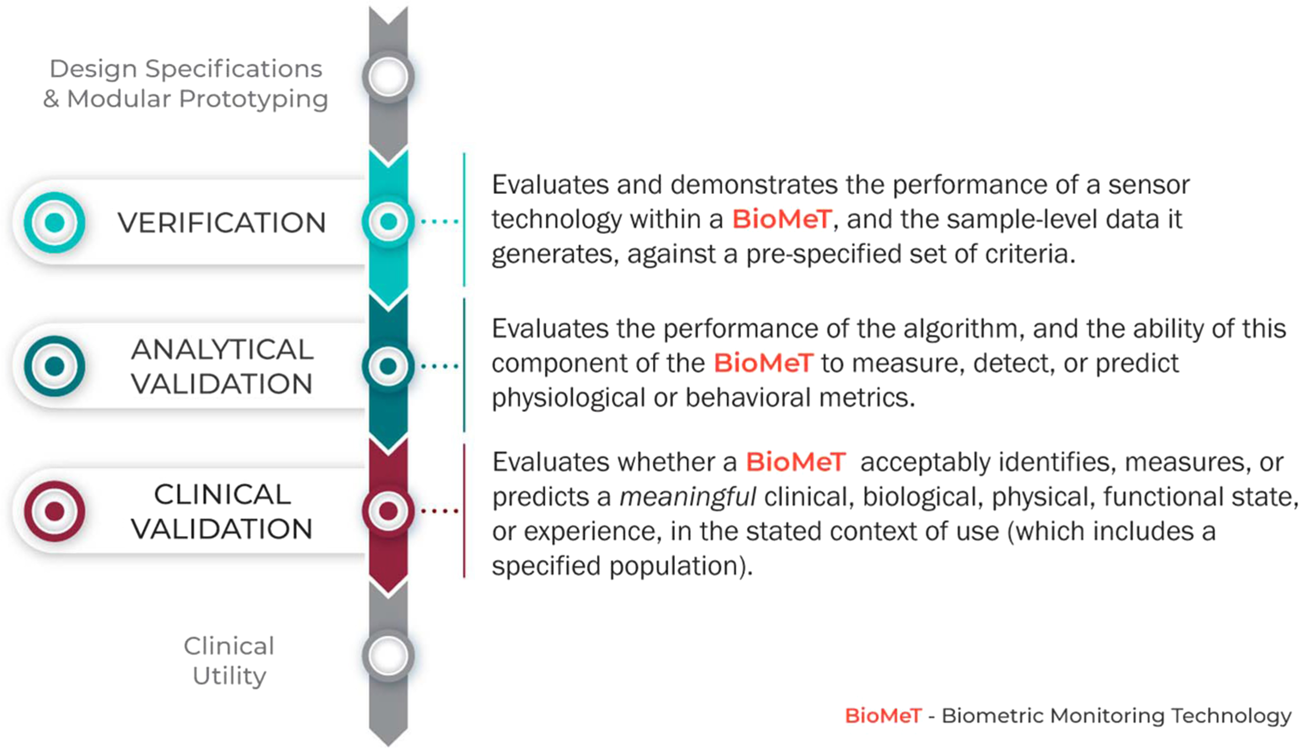
Verification, analytical validation, and clinical validation (V3): the foundation of determining fit-for-purpose for Biometric Monitoring Technologies (BioMeTs) | npj Digital Medicine

ISO 13485: Basics and How to Get Started (QMS for Medical Devices) | Process Street | Checklist, Workflow and SOP Software