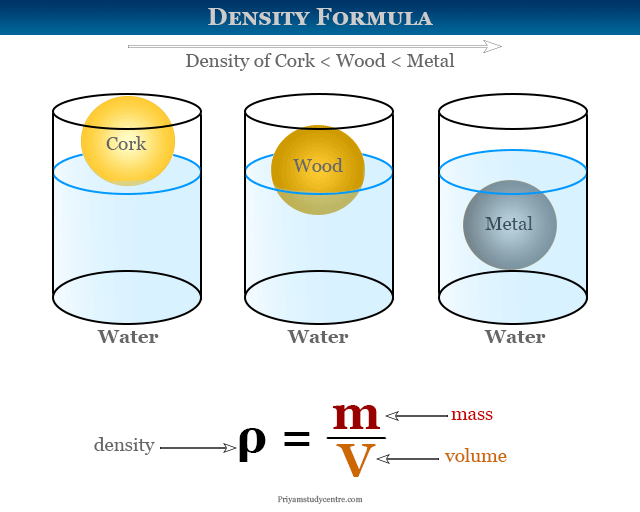
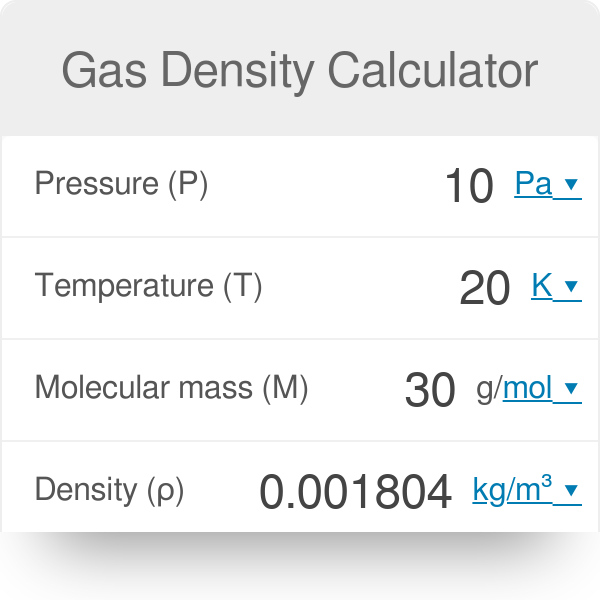
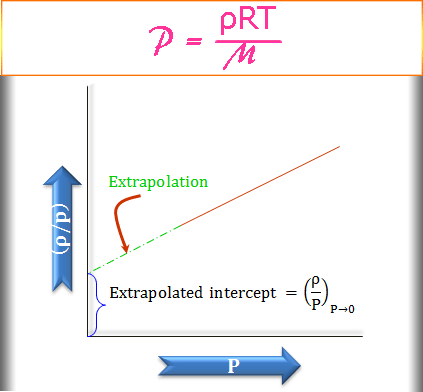
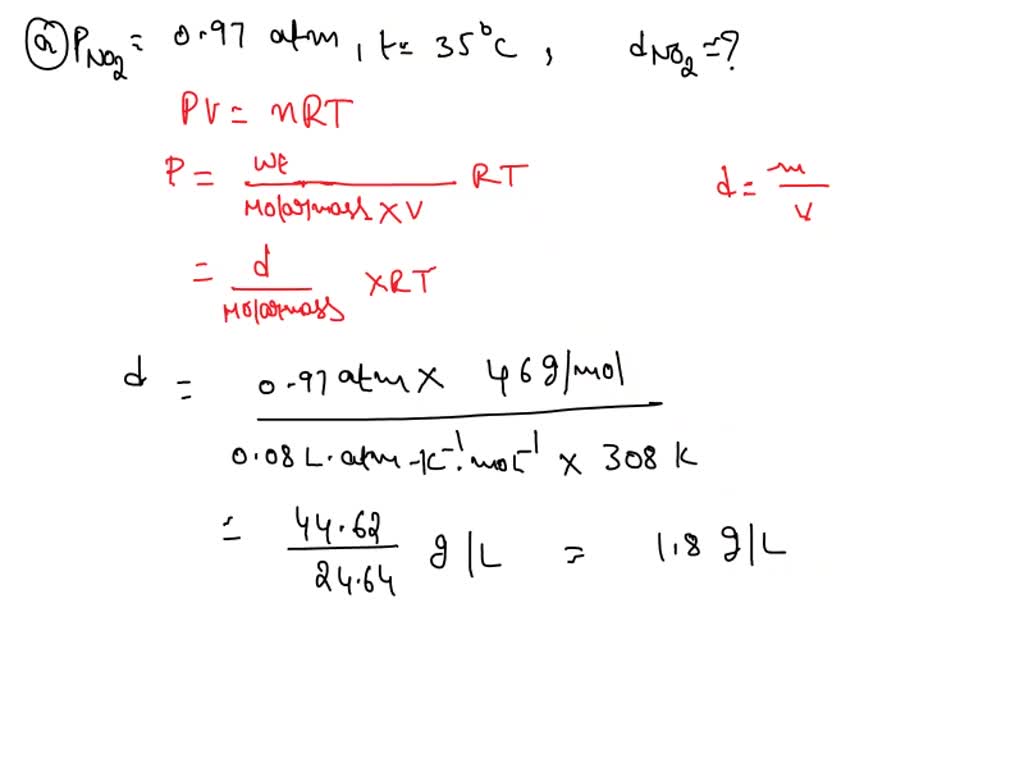How to Find Gas Density and Molar Mass at STP (Standard Temperature and Pressure) Example, Shortcut - YouTube

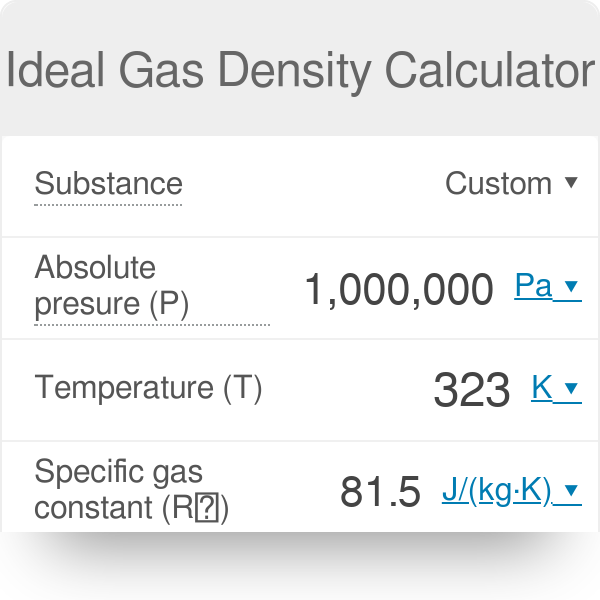
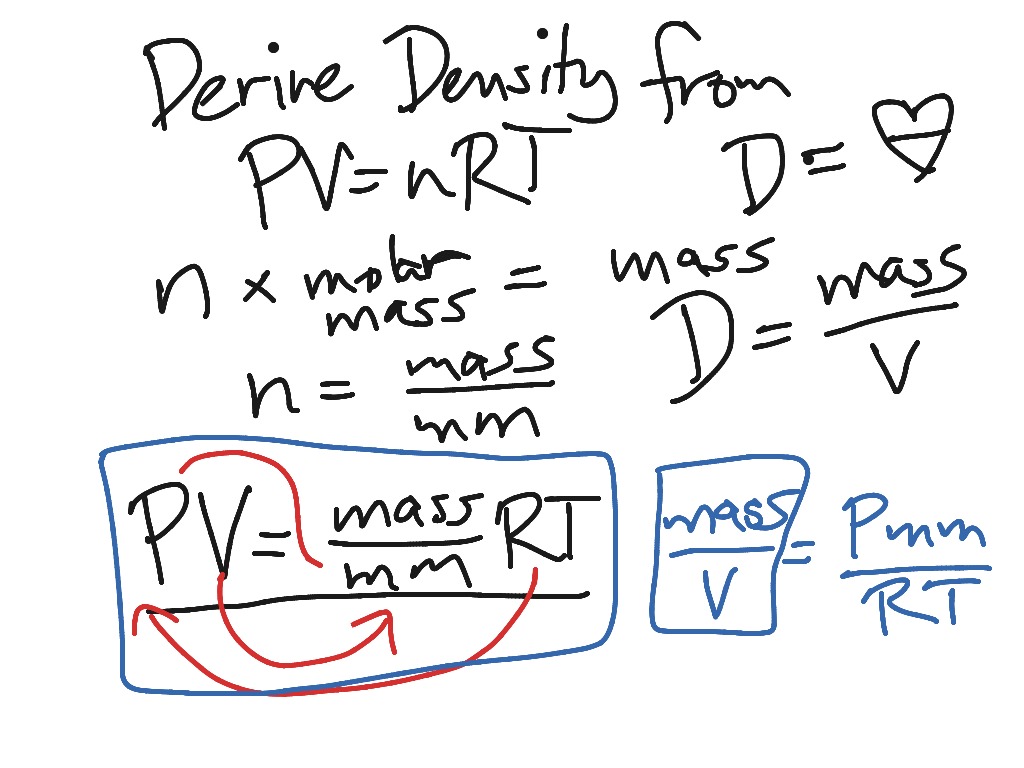
Gas Density Formula & Calculation | How to Find Density of a Gas - Video & Lesson Transcript | Study.com

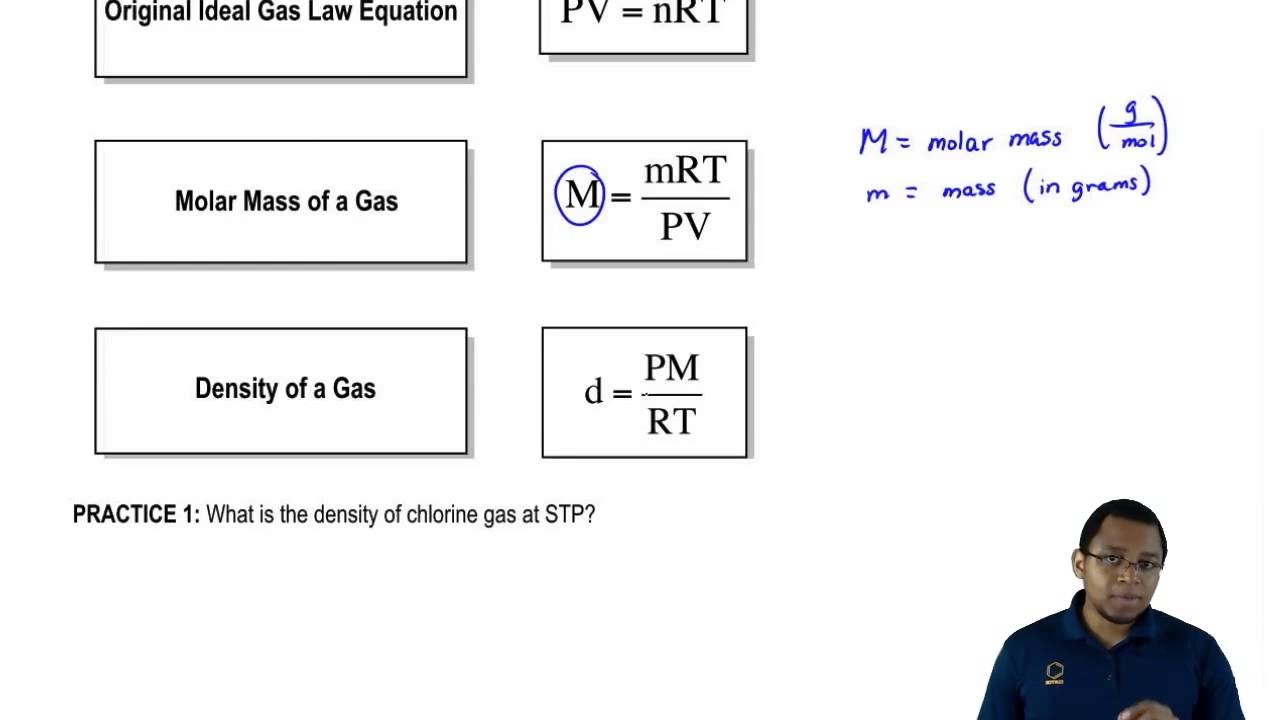
SOLVED: The density of an unknown gas at 20oC and 749 mm Hg is 1.31 g/L. Calculate the molar mass of the gas.