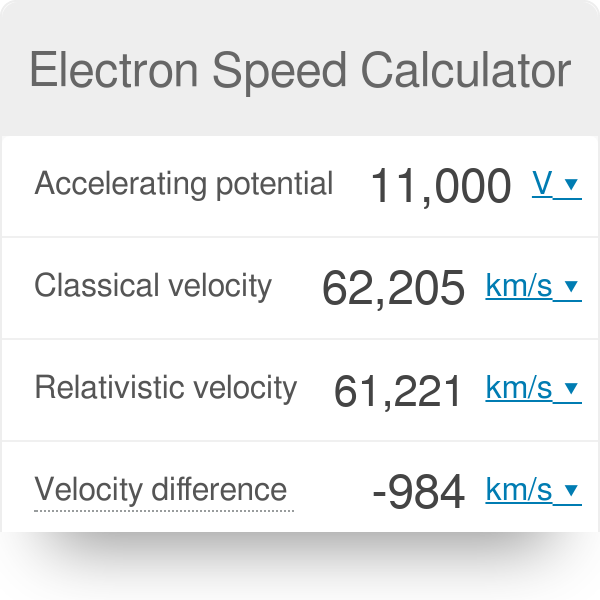
the velocity of an electron placed in 3rd orbit of h atom will be ans is 729 107 cm s its 10 raise to 7 give me its solution ukbsx511 -Chemistry - TopperLearning.com

An electron has a speed of 0.750c. (a) Find the speed of a proton that has the same kinetic energy - YouTube

Finding the Average Velocity of Electrons Emitted in a Photoelectric Experiment with Given Incident Light Frequency and Material Work Function | Physics | Study.com

An X-ray tube operates at `20k V`. Find the maximum speed of the electron strinking the anode , ... - YouTube
What is the formula for calculating the speed of electron if the kinetic energy of electron is given? - Quora

The average speed of electrons as functions of the applied potential... | Download Scientific Diagram

a) Using the Bohr's model calculate the speed of the electron in a hydrogen atom in the n = 1, 2, and 3 levels. (b) Calculate the orbital period in each of these levels.

The velocity of an electron in the first Bohr orbit of hydrogen atom is `2.19 xx 10^(6)ms^(-1)`. - YouTube
What is the formula for calculating the speed of electron if the kinetic energy of electron is given? - Quora
What is the formula for calculating the speed of electron if the kinetic energy of electron is given? - Quora